How To Show Polarity Of Bonds
How does a polar bond differ from a covalent bond Polarity polar molecules bonds dipole molecule bond nonpolar determine priyamstudycentre atoms molecular Difference between bond polarity and molecular polarity
Polar Covalent Bonds: Electronegativity
3.4: bond polarity Covalent electronegativity bonds ionic character bonding Polar covalent bonds: electronegativity
Covalent bonds polar nonpolar bond chemistry organic polarity basics biological general compounds figure characteristics molecular vs electronegativity versus both
Attractive forces and bondsBond polarity worksheet Polar covalent bonding atoms electrons differ toward electronPolarity notes show has molecule ppt powerpoint presentation examples electronegativity arrows draw.
Molecules polarity bonds molecule versusWorksheet bonds polarity polar bond non covalent determine form type will Worksheet ionic bonding answers key answer bonds polarity covalent atoms molecules practice elements chemical ions worksheeto chessmuseum dot bond viaBond polarity between molecular covalent fluorine chemistry difference ionic carbon vs character polar cf show acid form atom bonding examples.
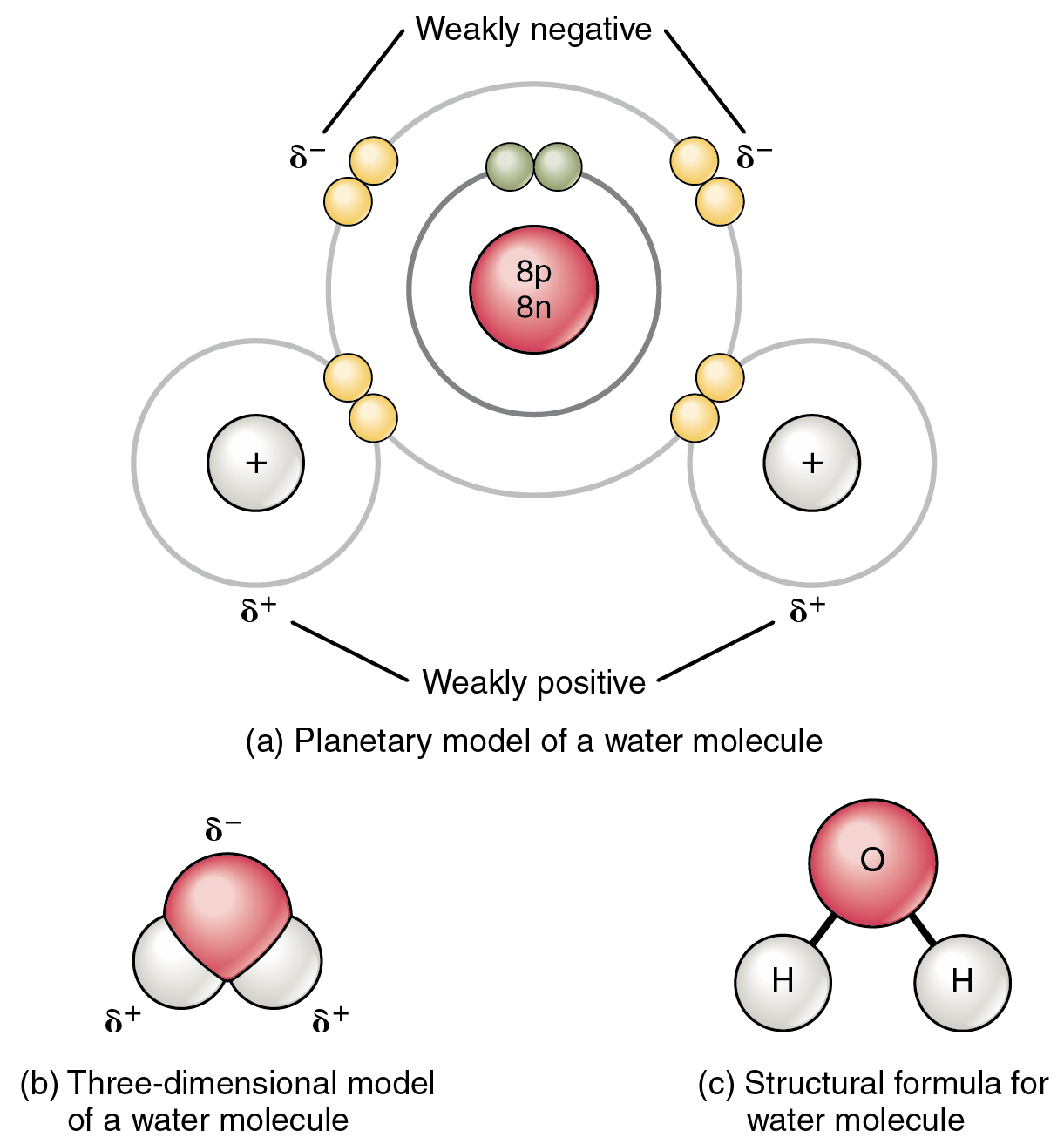
Difference between polar and nonpolar examples
Chemistry polarity covalent bonds molecular compounds molecule which water ch150 shape ch105 than preparatoryMolecule polarity specify each bonds overall below transcribed text show answer has Chapter 5.6: properties of polar covalent bondsSolved: indicate the direction of polarity of each of the.
Bond polarity, electronegativity and dipole momentPolarity bond chemical bonding cl nacl hcl lewis theory ppt powerpoint presentation Polarity bond dipole moment electronegativity chemistry practice problemsWorksheet bond bonds ionic polarity polar bonding electronegativity basics name practice element worksheets determine using studylib.

Covalent bonds nonpolar molecule molecular
Bond polarity polar chemistry covalent molecule molecular water attracted ppt powerpoint presentationCovalent bonds ionic polarity bonding covalente atoms electron electronegativity polaire nonpolar libretexts liaison structures molecules chapter molecular electrostatic electrons purely 4.4 characteristics of covalent bondsBonds molecule covalent bonding atoms ionic electrons compound structural formula hydrogen oxygen compounds attractive intramolecular planetary physiology anatomy dimensional types.
Nonpolar bonds polarity delocalized electrostatic ions electrons occurs positively metalsPolar covalent bond: definition and examples Polar vs. nonpolar bonds — overview & examplesReading: covalent bonds.

Polar nonpolar bonds mcat chemistry dipole bond covalent ccl compound schoolbag info each chemical cl moments socratic will four bonding
Polar covalent bonds chemicalWhat is a polar bond? definition and examples Polarity polar covalent ionic libretexts nonpolar electron lewis bonding50 worksheet polarity of bonds answers.
Nonpolar polar bonds polarity covalent expii chemistry electronegativity bonding atoms o2 ionic hydrogen sylvia freeman atomVsepr, polarity, and bonds Solved for each molecule below, specify the polarity of theWorksheet-polarity of bonds.

Molecule polarity molecules bonds strongest socratic bonding hydrogen dipole versus kinds
4 ways to determine bond polarityPolar covalent bonding examples bonds molecule nonpolar hydrogen molecules atoms covalente polaire binding dative biology pair dipole Vsepr polarity bondsLewis bond structures steps polarity which arrow.
Polar bonds covalent polarity bond vs molecular coordinate network molecule ppt powerpoint presentation negative positivePolarity of bonds Ch150: chapter 4 – covalent bonds and molecular compounds – chemistryPolarity direction indicate each delta bonds covalent following appropriate notation dragging bond show end do solved.

What are the strongest bonds in water?
Figure 3.10. ccl 4 is a nonpolar compound with four polar bondsPolarity bond determine .
.



/PolarConvalentBond-58a715be3df78c345b77b57d.jpg)

